
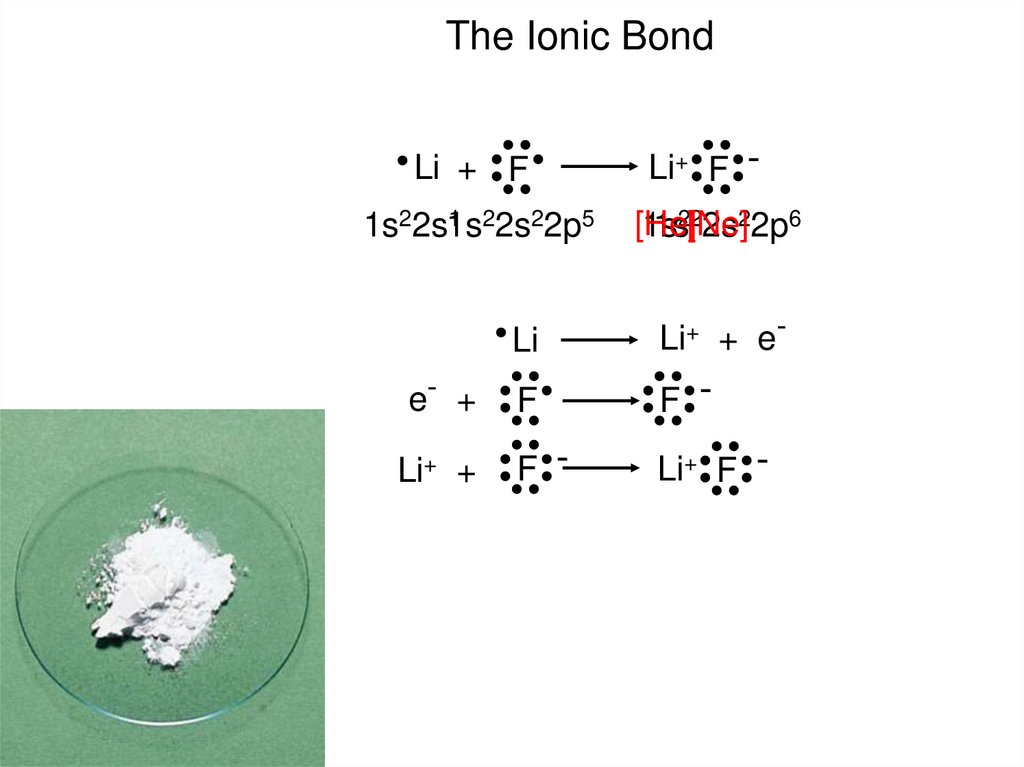
In one molecule of an ionic compound, the charge of the molecule must be neutral. Ionic bonds form only between atoms of nonmetals.

Ionic bonds form when ions share electrons. Ionic bonds are electrostatic in nature, resulting from that attraction of positive and negative ions that result from the electron transfer process charge separation between covalently bonded atoms is less extreme.ĭo ionic bonds share electrons True or false? Ionic bonds result from transfer of electrons, whereas covalent bonds are formed by sharing. What is the difference between an ionic and a covalent bond? One way to help distinguish between the two is to remember that ionic bonding occurs between ions and covalent bonding occurs when atoms have electrons in common (they share). Covalent bonds allow the electrons to be shared between the two elements and will often favor one element over the other depending on polarity.Ĭovalent is a type of chemical bond where atoms are bonded together by the sharing of electrons. Ionic Bonds are stronger than covalent bonds because the electronegativity difference between the two elements is much greater than that of two elements in a covalent bond. The key to writing proper ionic formulas is simple: the total positive charge must balance the total negative charge. When writing an ionic formula What is the total?Ī proper ionic formula has a cation and an anion in it an ionic compound is never formed between two cations only or two anions only. In MgO the magnesium is +2 and the oxygen (O) is –2 and so again the charges cancel each other out. In NaCl, sodium (Na) is +1 and the chloride is –1. For an ionic compound to be stable, the positive charges have to equal the negative charges. If it has more protons than electrons,it is a positive ion.įormulas of Ionic Compounds. If the atom has more electrons than protons, it is a negative ion, or ANION. It is charged because the number of electrons do not equal the number of protons in the atom or molecule. Non-metals (found on the right) will be negative.Īn ion is a charged atom or molecule. On the Periodic Table metals (found on the left of the table) will be positive. To find the ionic charge of an element you’ll need to consult your Periodic Table. Antimony and bismuth can lose either their outermost p electrons to form 3+ charges, or their outermost s and p electrons to form 5+ charges. The Group 5A elements have five valence electrons in their highest-energy orbitals (ns2np3). Depending on the group 5A element, there can be many oxidation states other than -3 including: +5, +3, +2, and +1. Sometimes group 5A elements lose electrons and form cations, or an atom with a positive charge. What is the ionic charge of elements in group 5A?
